Chronic venous insufficiency (CVI) in the legs, also known as varicose veins, is a relatively common condition, with worldwide prevalence ranging between 10% and 30%. Varicose veins can result in a number of symptoms, including pain, leg swelling, itching, and skin discoloration. Individuals with the condition may experience diminished quality of life and loss of work productivity. Finding treatment options that are effective and comfortable for the patient have been an area of interest in the endovascular community. 
A study by researchers in Verona, Italy, found that endovenous mechanochemical ablation (MOCA) treatment with the ClariVein® OC Infusion Catheter was associated with a good occlusion rate, comparable with other techniques, including thermal techniques, without major complications. This is one of the first reports in literature for MOCA treatment of symptomatic varicose veins with 5-year results. With little long-term data pertaining to this type of treatment, the aim of the study was to evaluate long-term occlusion rates.
This retrospective single-center study was published in Journal of Vascular Surgery: Venous and Lymphatic Disorders. Mirandola et al. treated 395 primary, symptomatic, unilateral, incompetent varicose saphenous veins in an outpatient setting. Of these patients, none were treated bilaterally in the same session. The majority of vessels treated were great saphenous veins (92.3%), and the others were small saphenous veins. Procedures were performed with the ClariVein OC and polidocanol 2% in liquid form.
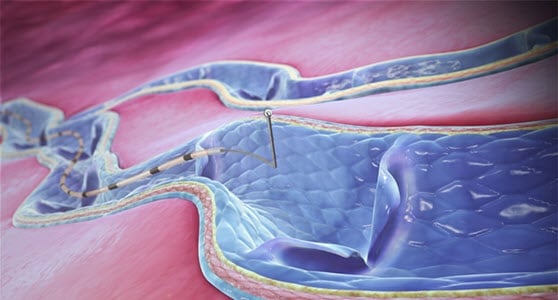 A total of 329 patients were available for follow up, each treated between September 2012 and September 2017. Average follow-up time was 20 ± 18 months (range, 6-60 months). Results showed a technical success of 99.5%. Treatment was incomplete in two patients due to vein spasm, leading to catheter damage and inability to infuse the sclerosant. Follow up consisted of clinical evaluation and duplex ultrasound scan performed at 1 week, 1 month, 6 months, 1 year, and then once every year.
A total of 329 patients were available for follow up, each treated between September 2012 and September 2017. Average follow-up time was 20 ± 18 months (range, 6-60 months). Results showed a technical success of 99.5%. Treatment was incomplete in two patients due to vein spasm, leading to catheter damage and inability to infuse the sclerosant. Follow up consisted of clinical evaluation and duplex ultrasound scan performed at 1 week, 1 month, 6 months, 1 year, and then once every year.
The overall survival rate free from recanalization was 92.4%. Anatomic success was 94% at 1 year, 91% at 2 years, 88% at 3 years, 88% at 4 years, and 84% at 5 years. The follow up at 5 years included 23 patients, five of whom presented with recanalization. The authors note that in this series, the veins completely disappeared on duplex ultrasound scan in 36% of cases.
It is interesting to note that after 89 patients, the researchers changed the way in which the sclerosant was injected. Beginning the infusion of sclerosant at point of mechanical pullback (protocol 2) produced higher occlusion rates compared to traditional ClariVein OC technique (protocol 1) with delayed infusion of sclerosant. Protocol 2 resulted in a change in cumulative survival rate free from recanalization from 92% to 97% (P ¼ 0.013).
 Fig 4. Difference in anatomic success between the two different protocols.
Fig 4. Difference in anatomic success between the two different protocols.
The researchers concluded that the ClariVein OC was associated with a good occlusion rate and was comparable with other techniques, including thermal techniques, without major complications. 
Varicose veins occur when vessels are unable to pump blood back to the heart, causing the blood to pool, resulting in vessel enlargement over time. Under normal conditions, blood moves from the heart to the legs through the arteries and back to the heart through the veins. To accomplish this, the veins rely on surrounding muscles and a collection of valves to prevent blood from flowing backward. When these muscles and valves weaken or fail to work, blood begins to gather in the vein instead of returning to the heart.
In addition to pain, swelling, itching, and skin discoloration, other symptoms of varicose veins include:
- Twisted and bulging veins
- Aching, burning, throbbing and muscle cramping
- Leg heaviness and fatigue
The ClariVein OC alleviates these symptoms by combining the power of a specialty infusion catheter with a rotating wire tip designed for the controlled 360-degree dispersion of physician-specified agents inside the targeted vessel. Requiring only a pin-sized entry through the skin, treatment with the ClariVein OC takes very little time, uses no heat or tumescent anesthesia, and creates minimal discomfort to the patient.
Learn more about the clinically proven ClariVein OC and how it can change the lives of your patients. Visit ClariVein.com or contact our Customer Support for more information.
ARTICLE REFERENCED:
Mirandola M et al. 2020. “An Italian Experience with Mechanochemical Ablation of the Saphenous Vein Since 2012.” J Vasc Surg Venous Lymphat Disord 8, no 6 (Nov): 999–1005. PMID: 32179039.