Varicose veins are a common condition worldwide. Currently estimated to affect up to 3 in 10 adults,1 varicose veins are responsible for significantly diminished quality of life with consequent healthcare costs.2
To treat varicose veins, endovenous thermal ablation has been considered the gold standard of care—but this technique is not without patient discomfort. In recent years, new therapies have been developed and research implemented in an effort to improve the treatment experience for patients with varicose veins.
A multi-centre randomized controlled trial2 by researchers at Imperial College London and London North West Hospitals NHS Trust found that compared with radiofrequency ablation—a type of endovenous thermal ablation—treatment with minimally invasive ClariVein OC was less painful and produced similar short-term results as well as quality of life and safety outcomes.
The study,2 published in Phlebology, recruited 170 patients and randomly assigned them to undergo either radiofrequency ablation or treatment with ClariVein OC. Researchers examined pain scores using a Visual Analogue Scale (VAS) and number scale (0 ̶ 10) during treatment. Patients were followed up at one and six months with clinical scores, quality of life scores, and ultrasound assessment of the treated leg.
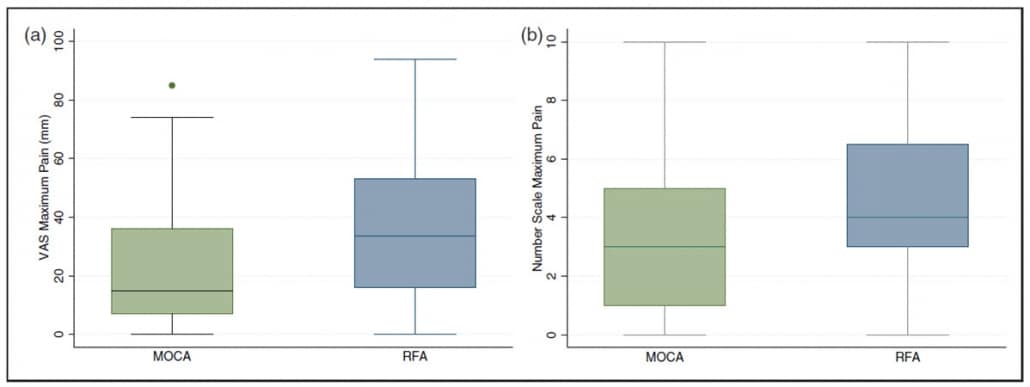
Results2 showed that patients in the ClariVein OC group experienced significantly less pain during the procedure by both VAS (ClariVein OC median 15 mm [IQR 7–36 mm] vs. radiofrequency ablation 34 mm [IQR 16–53 mm], p = 0.003) and number scale (ClariVein OC median 3 [IQR 1–5] vs. radiofrequency ablation 4 [IQR 3–6.5], p = 0.002).
Data2 also showed that average pain scores were significantly less in the ClariVein OC group (VAS median 10 mm [3–25 mm] vs. 19.5 mm [9–38 mm], p = 0.003, Mann-Whitney, and number scale median of 2 [0.5–4] versus 3 [2–5], p = 0.004, Mann Whitney).
Occlusion rates, clinical severity scores, disease specific and generic quality of life scores were similar between groups at one and six months.2 Overall, patients returned to work and normal activities at a median of two days (IQR 2–7 and 1–6, respectively). Adverse events included two deep vein thromboses, one in each group.
The researchers concluded2 that treatment for varicose veins is less painful with ClariVein OC than radiofrequency ablation with similar short-term technical, quality of life, and safety outcomes. Clinical outcomes and time returning to work and normal activities were all similar. These findings support other data comparing patient pain between ClariVein OC and radiofrequency ablation3 as well as published five-year follow-up data examining ClariVein OC’s long-term efficacy.4
Results2 showed that patients in the ClariVein OC group experienced significantly less pain during the procedure by both VAS (ClariVein OC median 15 mm [IQR 7–36 mm] vs. radiofrequency ablation 34 mm [IQR 16–53 mm], p = 0.003) and number scale (ClariVein OC median 3 [IQR 1–5] vs. radiofrequency ablation 4 [IQR 3–6.5], p = 0.002).
Data2 also showed that average pain scores were significantly less in the ClariVein OC group (VAS median 10 mm [3–25 mm] vs. 19.5 mm [9–38 mm], p = 0.003, Mann-Whitney, and number scale median of 2 [0.5–4] versus 3 [2–5], p = 0.004, Mann Whitney).
Occlusion rates, clinical severity scores, disease specific and generic quality of life scores were similar between groups at one and six months.2 Overall, patients returned to work and normal activities at a median of two days (IQR 2–7 and 1–6, respectively). Adverse events included two deep vein thromboses, one in each group.
The researchers concluded2 that treatment for varicose veins is less painful with ClariVein OC than radiofrequency ablation with similar short-term technical, quality of life, and safety outcomes. Clinical outcomes and time returning to work and normal activities were all similar. These findings support other data comparing patient pain between ClariVein OC and radiofrequency ablation3 as well as published five-year follow-up data examining ClariVein OC’s long-term efficacy.4
Varicose veins can be unsightly and for some patients cause discomfort. The underlying cause of varicose veins is venous reflux disease, which occurs when the valves inside veins become weakened or damaged. When this happens, blood flows backwards and collects inside the vein, causing it to enlarge. Varicose veins are most common in the legs and feet. Left untreated, venous reflux disease can progress to create other more serious circulatory and skin problems.
To treat varicose veins, radiofrequency ablation uses imaging guidance to navigate a small catheter into the diseased vein to deliver heat, causing vessel walls to collapse and close. Although considered effective, radiofrequency ablation requires the use of tumescent anaesthesia which involves multiple needle injections.
In contrast, ClariVein OC combines the power of a specialty infusion catheter with a rotating wire tip designed for the controlled 360-degree dispersion of physician-prescribed medication inside the targeted vessel. Requiring only a pin-sized entry through the skin, treatment with the ClariVein OC takes very little time, uses no heat or tumescent anaesthesia, and creates minimal discomfort to the patient.
Learn more about the clinically proven ClariVein OC and how it can change the lives of your patients. Visit ClariVein.com or contact our Customer Support for more information.
IMPORTANT
This product page is intended only for an audience outside of the United States, as ClariVein OC is not approved, cleared, or available for sale in the United States. This product is not approved, cleared, or available for sale or use in the United States, and may not be approved, cleared, or available for sale or use in other countries.
Please check with your country representative to determine which ClariVein catheters are available in your country.
Please consult the product Instructions for Use (IFU) for indications, contraindications, potential complications, warnings, precautions, and directions for use.
References
- NHS Inform. 13 February 2020. “Varicose Veins.” https://www.nhsinform.scot/illnesses-and-conditions/heart-and-blood-vessels/conditions/varicose-veins
- Lane et al. 2016. “A Multi-Centred Randomised Controlled Trial Comparing Radiofrequency and Mechanical Occlusion Chemically Assisted Ablation of Varicose Veins—Final Results of the Venefit versus ClariVein for Varicose Veins Trial.” Phlebology 32, no. 2 (March): 89–98. PMID: 27221810.
- Vun et al. 2015. “Lower Pain and Faster Treatment with Mechanico-Chemical Endovenous Ablation Using ClariVein®.” Phlebology 30, no 10 (Dec): 688–692. PMID: 25300311.
- Mirandola et al. 2020. “An Italian Experience with Mechanochemical Ablation of the Saphenous Vein Since 2012.” J Vasc Surg Venous Lymphat Disord 8, no 6 (Nov): 999–1005. PMID: 32179039.