Categories
Procedure
Embosphere Microspheres temporarily compress up to 33% for smooth microcatheter passage. Once through the microcatheter, they return to their original spherical shape and stated diameter for predictable, reliable delivery.1
Embosphere Microspheres show a direct correlation between the level of arterial occlusion and the size of the particles used, allowing for consistent and reliable targeted occlusion.2,3
Embosphere Microspheres have been proven:
• In more than 130 pivotal clinical articles3
• In over 8,000 procedures reported in clinical studies
• In over 25 years of clinical experience
• In more than 800,000 patients worldwide
The hydrophilic surface and spherical shape of Embosphere Microspheres prevent aggregation in the catheter lumen and vasculature.1
Embosphere Microspheres’ rebound capabilities help maintain a spherical shape for predictable distribution & occlusion. In contrast, Embozene® Microspheres show a higher in vivo deformation, resulting in unpredictable occlusion.4
Embosphere Microspheres’ elastic properties allow temporary compression of up to 33% to facilitate smooth microcatheter passage (left image).1 Once through the microcatheter, Embosphere Microspheres return to their original spherical shape and stated diameter (right image) for predictable, targeted delivery.1
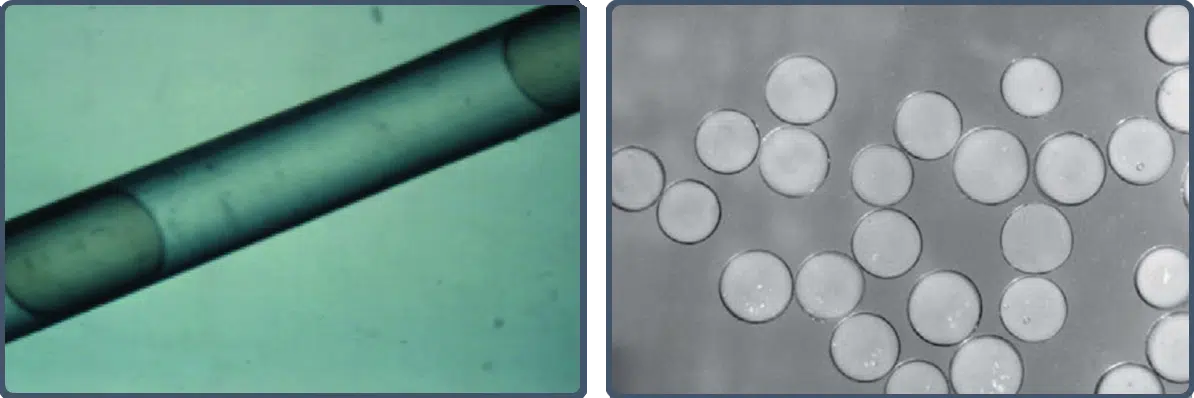
Embosphere Microspheres are biocompatible and nonresorbable with cell adhesion properties that enable complete and durable mechanical occlusion.1
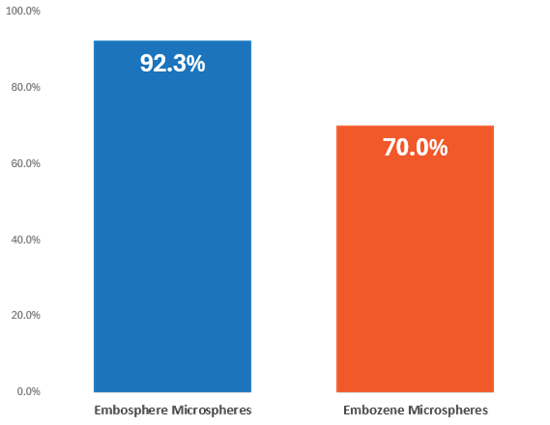 Embosphere Microspheres offer high total fibroid infarction rates of up to 92.3%, compared to infarction rates for competitive devices of up to 70.0%.5,6
Embosphere Microspheres offer high total fibroid infarction rates of up to 92.3%, compared to infarction rates for competitive devices of up to 70.0%.5,6
UFE rates are defined differently, results are from different studies and may vary in head-to-head comparison. Graphics are for illustration purposes only.
REFERENCES
1. Laurent et al. 1996. “Trisacryl Gelatin Microspheres for Therapeutic Embolization, I: Development and In Vitro Evaluation.” Am J Neuroradial. 17, no. 3 (Mar):533-40
2. Pelage et al. 2002. “Uterine Artery Embolization in Sheep: Comparison of Acute Effects with Polyvinyl Alcohol Particles and Calibrated Microspheres.” Radiology 224 no. 2 (Aug): 436-45. doi: 10.1148/radiol.2242010847.
3. Data on file.
4. Verret et al. 2011. “The Arterial Distribution of Embozene and Embosphere Microspheres in Sheep Kidney and Uterus Embolization Models.” J Vasc Interv Radiol. 22, no. 2 (Feb):220-8. doi:10.10106/j.jvir.2010.10.021.
5.Maclean et al. 2021. “A Comprehensive Cohort Study Comparing a Novel, Spherical, Resorbable Particle Against Five Established Embolic Agents for Uterine Fibroid Embolisation.” Clin Radiol 76, no. 6 (June) 452-57. Accessed February 07, 2025. doi: 10.1016/j.crad.2021.01.012. Epub 2021 Feb 23.
6. Siskin et al. 2008. “Leiomyoma Infarction After Uterine Artery Embolization: A Prospective Randomized Study Comparing Tris-Acryl Gelatin Microspheres Versus Polyvinyl Alcohol Microspheres.” J Vasc Interv Radiol 19, no. 1 (January): 58 ̶ 65. Accessed October 23, 2024. doi: 10.1016/j.jvir.2007.08.034.
![]()
Additional Documents
Embosphere Microspheres (vial) with GAE indication – MDR – IFU (European version)
Embosphere Microspheres (syringe) with GAE indication – MDR – IFU (European version)
409050001_001 ID042425