Categories
Procedure
Predictable, Flow-Directed Targeting
QuadraSphere Microspheres are calibrated, hydrophilic and compress up to 70% to facilitate smooth microcatheter delivery.* Once through the microcatheter, they rebound to their stated spherical shape with a consistent cross-sectional diameter for predictable, flow-directed targeting.
Targeted Occlusion
The in vivo median diameter of occluded arteries is significantly different between sizes of QuadraSphere Microspheres,* providing targeted occlusion.
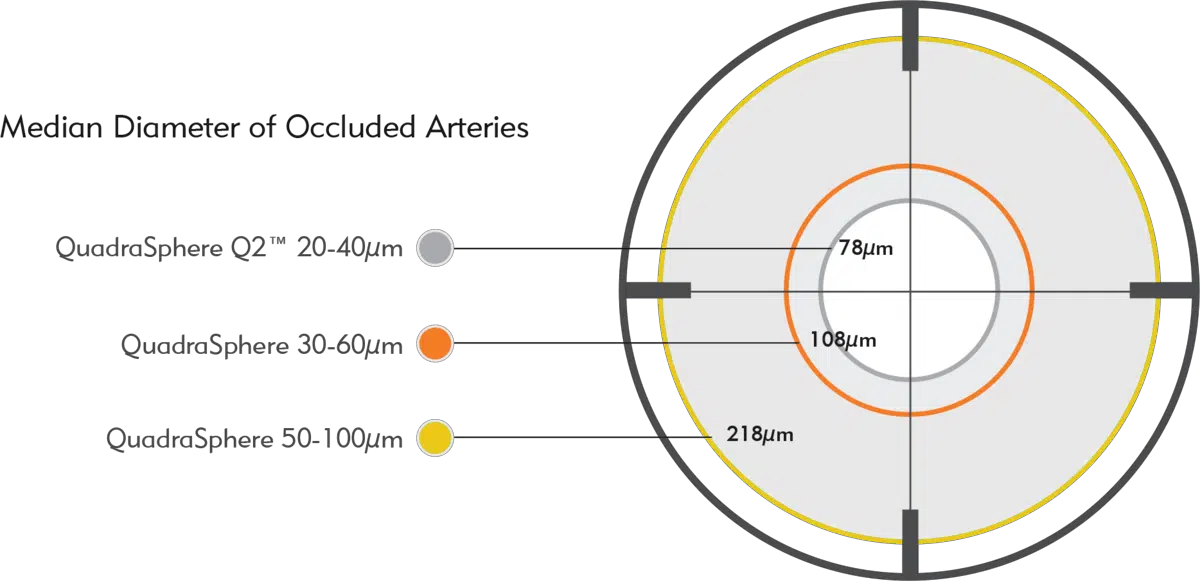
QuadraSphere Q2 Microspheres demonstrate a median occlusion of 78 µm, a statistically significant difference compared to larger sizes of QuadraSphere.
*Swine liver embolization model study performed for Merit Medical.* QuadraSphere Microspheres prepared in 50/50 mixture of saline/contrast.
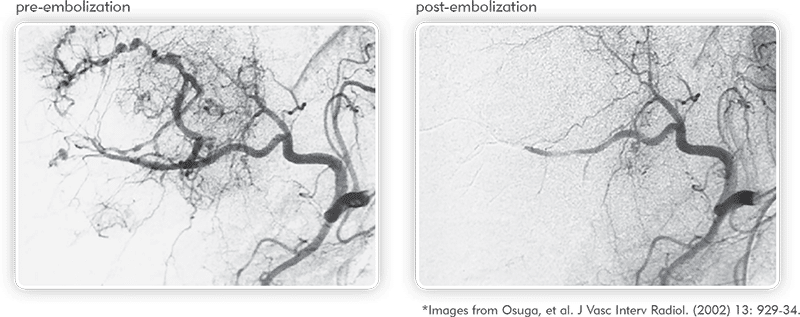
Angiographies of hypervascularized tumor pre- and post-embolization illustrate the ability of QuadraSphere Microspheres to perform a targeted embolization.1
Experience the Power of Conformability
QuadraSphere Microspheres are soft and easily conform to the vessel lumen for complete occlusion:2,3
Established
QuadraSphere Microspheres are a recognized and accepted platform for targeted liver embolization.
QuadraSphere Microspheres are indicated for embolization of hypervascularized tumors including hepatoma, and peripheral arteriovenous malformations.
Super-Absorbent Polymer
Referred to as super-absorbent polymer (SAP) microspheres, QuadraSphere Microspheres are packaged dry, but once exposed to aqueous-based media, they swell to approximately 4x the size printed on the product label.*
REFERENCES
* Data on file.
1. Osuga et al. 2001. “Transarterial Embolization for Large Hepatocellular Carcinoma with Use of Superabsorbent Polymer Microspheres: Initial Experience.” J Vasc Interv Radiol 2002; 13:929–934.
2. Bilbao JI et al. Comparative study of four different spherical embolic particles in an animal model: a morphologic and histologic evaluation. J Vasc Interv Radiol 2008 Nov;19(11):1625-38.
3. De Luis et al. 2007. “In Vivo Evaluation of a New Embolic Spherical Particle (HepaSphere) in a Kidney Animal Model.” Cardiovasc Intervent Radiol 31, no. 2 (Mar–Apr): 367–76. doi: 10.1007/s00270-007-9240-1.
409051001_001 ID 051425