Categories
Procedure

The SAVI Brachy applicator is gently inserted in a closed position into the tumor cavity through a small incision, a procedure that is usually done in the physician’s office.
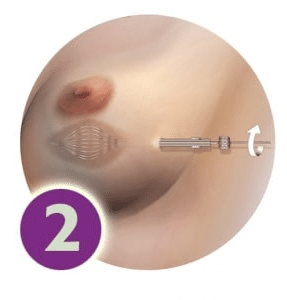
The catheters are gently expanded to conform to the shape of the cavity. This is done only once, at the beginning of treatment.
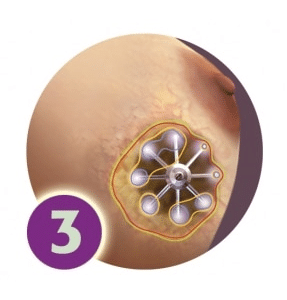
Once the catheter is in place, a radiation oncologist delivers treatment twice a day for 2-5 days. The ends of the catheters are connected to a computerized delivery system that painlessly delivers a tiny radioactive seed into each tube. The radiation source is completely removed after each treatment – no radiation remains in the body between treatments.
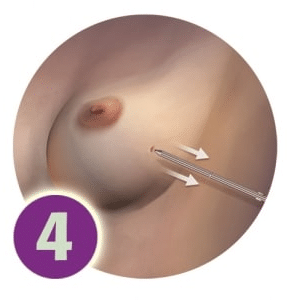
After the last radiation treatment, the physician will close the device and remove it through the same incision into which it was originally inserted.
FEATURED STUDY:
Three-Fraction Accelerated Partial Breast Irradiation (APBI) Delivered With Brachytherapy Applicators Is Feasible and Safe: First Results From the TRIUMPH-T Trial
200 patients enrolled August 2015 – August 2017
Int J Radiat Oncol Biol Phys. 2019;104(1):67-74. doi:10.1016/j.ijrobp.2018.12.050.
Primary Objective:
Toxicity @ 2 yrs
Secondary Objectives:
Cosmesis @ 2 yrs.
Local control @ 3 yrs:
Ultrashort breast brachytherapy is dosimetrically feasible and can be delivered with excellent short-term tolerance and low toxicity.
Peer Reviewed: L.E. Simon et al. (2022). Retrospective review of three-fractioned accelerated partial breast irradiation, Brachytherapy, https://doi.org/10.1016/j.brachy.2022.02.002
![]() Download Study
Download Study
Case Review: Yashar (2019). SAVI Brachytherapy with TRIUMPH-T 2-day Treatment Regimen.
![]() Download Case Study
Download Case Study
Peer Reviewed: Wilkinson JB, Chen PY, Wallace MF, et al. (2018). Six-Year Results From a Phase I/II Trial for Hypofractionated Accelerated Partial Breast Irradiation Using a 2-Day Dose Schedule. Am J Clin Oncol. 2018;41(10):986-991. doi:10.1097/COC.0000000000000402
https://pubmed.ncbi.nlm.nih.gov/28787281/
![]() Preview
Preview
Peer Reviewed: Yashar C, Attai D, Butler E, et al. (2016). Strut-based accelerated partial breast irradiation: Report of treatment results for 250 consecutive patients at 5 years from a multicenter retrospective study. Brachytherapy. 2016;15(6):780-787. doi:10.1016/j.brachy.2016.07.002
https://pubmed.ncbi.nlm.nih.gov/27528591/
![]() Preview
Preview
Peer Reviewed: Rehman S, Agarwal R, Ochoa L, et al. (2016). Prospective analysis of toxicity in patients treated with strut-adjusted volume implant for early-stage breast cancer. Brachytherapy. 2016;15(5):625-630. doi:10.1016/j.brachy.2016.04.008
https://pubmed.ncbi.nlm.nih.gov/27263058/
![]() Preview
Preview
Peer Reviewed: Mooney KE, Altman MB, Edward S, et al. (2016). Accelerated partial breast irradiation dosimetric criteria for the strut-adjusted volume implant. Brachytherapy. 2016;15(5):616-624. doi:10.1016/j.brachy.2016.05.002
https://pubmed.ncbi.nlm.nih.gov/27349857/
![]() Preview
Preview
Strnad et al. (2015). 5-year results of accelerated partial breast irradiation using sole interstitial multicatheter brachytherapy versus whole-breast irradiation with boost after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: a randomised, phase 3, non-inferiority trial. 2016;387(10015):229-238, Jan 16, 2016. doi:https://doi.org/10.1016/S0140-6736(15)00471-7
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(15)00471-7/fulltext
![]() Preview
Preview
Looking for more? Find Pre-2015 studies here >>
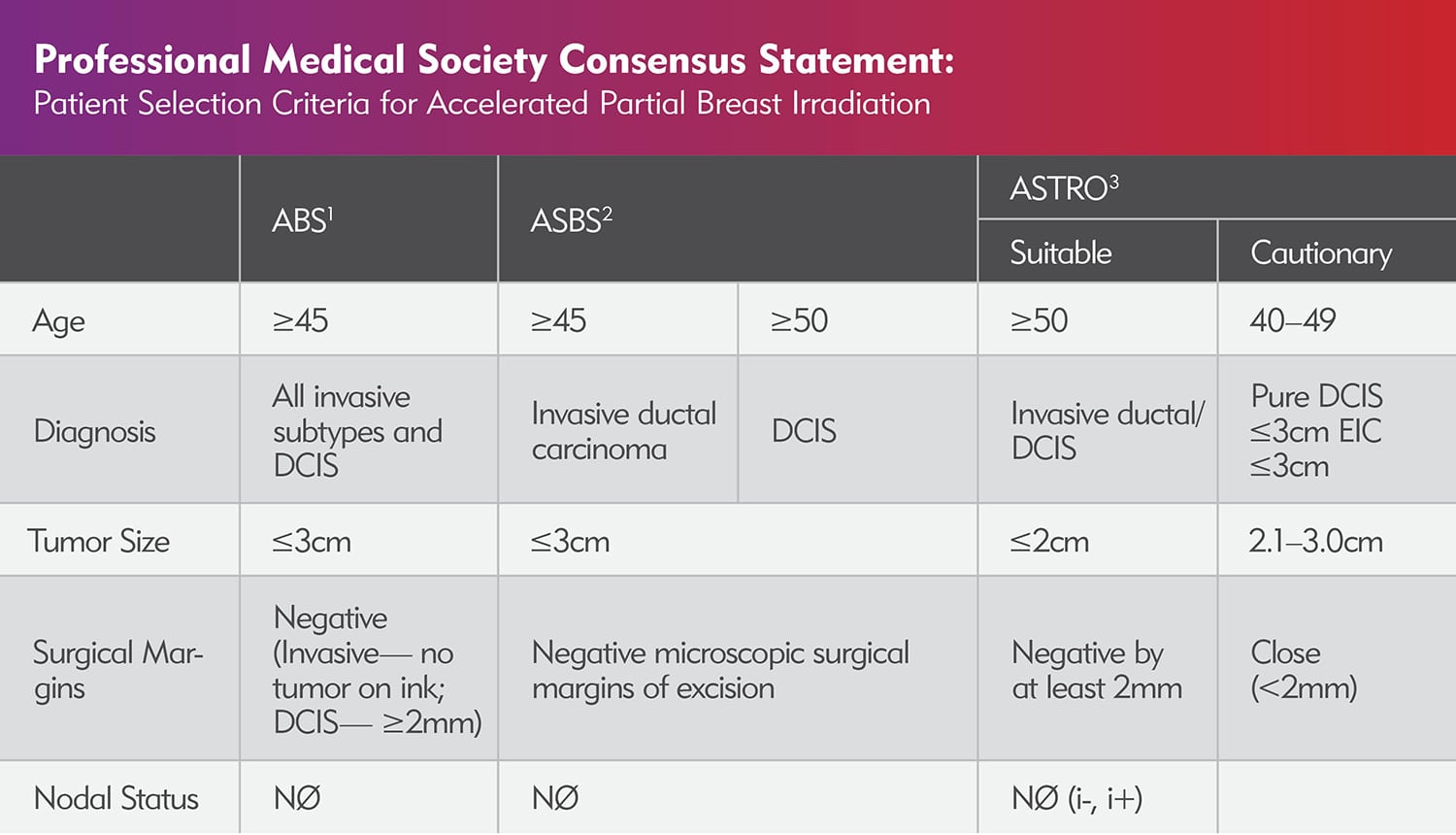 Patient Selection Criteria PDF
Patient Selection Criteria PDF
Shah C, Vicini F, Shaitelman SF, et al. (2017). The American Brachytherapy Society consensus statement for accelerated partial-breast irradiation. Brachytherapy. 2018;17(1):154-170. doi:10.1016/j.brachy.2017.09.004 https://pubmed.ncbi.nlm.nih.gov/29074088/
The American Society of Breast Surgeons (2011). Consensus statement for accelerated partial breast irradiation. https://www.breastsurgeons.org/docs/statements/Consensus-Statement-for-Accelerated-Partial-Breast-Irradiation.pdf
American Society Radiation and Oncology (2016). Consensus Statement on Partial Breast Irradiation. https://www.practicalradonc.org/article/S1879-8500(16)30184-9/fulltext
REFERENCES