Categories
Procedure

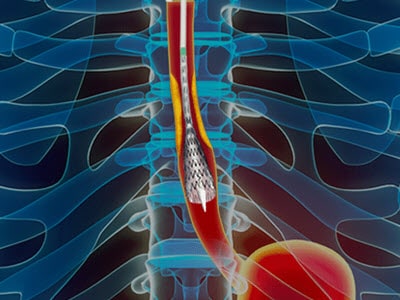
Merit Endotek pioneered the world’s first fully covered, FDA-approved esophageal stent. Learn how we’re expanding and improving that legacy with a complete range of innovative, next-generation gastrointestinal solutions.

Find out why experienced clinicians trust Merit Endotek’s family of fully-covered, laser-cut AERO tracheobronchial stents and advanced over-the-wire and direct visualization delivery systems to deliver consistent, reliable results.