Categories
-
Access
-
Accessories
-
Balloons
-
Biopsy
-
Cardiac Rhythm Management
-
Catheters
-
Compression
-
Critical Care
-
Dialysis Access
-
Drainage
-
Electrophysiology
-
Embolics & Delivery Systems
-
GERD Treatment
-
Guide Wires
-
Inflation Devices
-
Interventional Spine
-
Kits and Packs
-
Monitoring Devices
-
Needles
-
Oncology
-
Safety
-
Snares
-
Stents
-
Syringes
Procedure

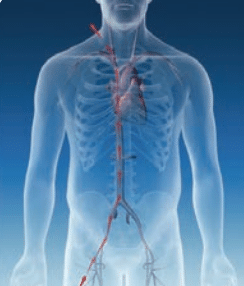 RESTORE ACCESS
RESTORE ACCESS RELIABLE PROCEDURE
RELIABLE PROCEDURE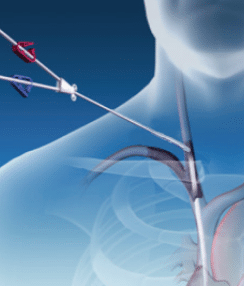 REDUCE COSTS
REDUCE COSTS