Categories
Procedure
Compared to other catheters, HeRO Graft has:
Fewer Infections: 69% reduced infection rate compared with catheters1
Up to 87% cumulative patency at 2 years1,2
A 23% average savings per year compared with catheters3
HeRO Graft may be right for your or your patient if:
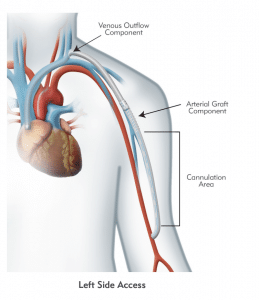
HeRO Graft (Hemodialysis Reliable Outflow) is the ONLY fully subcutaneous AV access solution clinically proven to maintain long-term access for hemodialysis patients with central venous stenosis. HeRO Graft is classified by the FDA as a graft, but differs from a conventional AV graft since it has no venous anastomosis. It consists of two primary components:
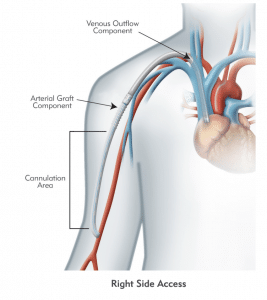
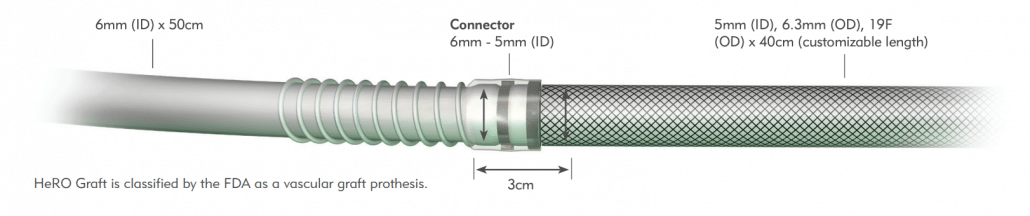
The HeRO Graft Arterial Graft Component has a 6mm inner diameter (ID), 7.4mm outer diameter (OD), and is 53cm long, inclusive of the connector. It consists of an ePTFE hemodialysis graft with PTFE beading to provide kink resistance near the proprietary titanium connector. The titanium connector attaches the Arterial Graft Component to the Venous Outflow Component. The Arterial Graft Component is cannulated using standard technique according to KDOQI guidelines.
The HeRO Graft Venous Outflow Component has a 5mm ID, 19F (6.3mm) OD, and is 40cm long. It consists of radiopaque silicone with braided nitinol reinforcement (for kink and crush resistance) and a radiopaque marker band at the distal tip.
See the HeRO Graft Instructions for use.
References
Product Documents
HeRO Graft – Care and Cannulation