Categories
Procedure
Expand your range of treatment options
Any patient with focalized pain from a metastatic spinal tumor may be a candidate for t-RFA, several specific patient groups will likely benefit most from the procedure.
Patients Who Benefit from RF Spine Tumor Ablation with The STAR™ Tumor Ablation System1-4
Patients with focal pain from metastatic spinal tumor(s) may be a candidate for treatment. However, patients that are ideally suited include those with:
“There are patients participating in some chemotherapy clinical trials for their primary cancer which do not allow radiation therapy treatments [while participating in the study]. The STAR™ procedure is an excellent option for these patients because it allows us to treat the localized area and relieve the pain without the need to stop the ongoing therapy they receive on the trial.”
Jayson Brower, M.D.*
Interventional Radiologist
Inland Imaging, Spokane WA
For localized pain, such as that associated with painful metastatic spinal lesions, the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for Adult Cancer Pain indicate that radiofrequency ablation should be a considered treatment option.
Oncologists and cancer care team members recognize the fast and durable pain relief patients achieve with minimally invasive targeted radiofrequency ablation (t-RFA).
The STAR™ Tumor Ablation System, t-RFA delivers meaningful pain relief and localized tumor destruction in a single treatment, often in an outpatient setting.
The STAR™ System provides physicians the control to create site-specific ablation zones and monitor real-time ablation zone development within the vertebral body.
t-RFA expands the range of treatment options for patients with painful metastatic spinal tumors as it is compatible with systemic therapies.
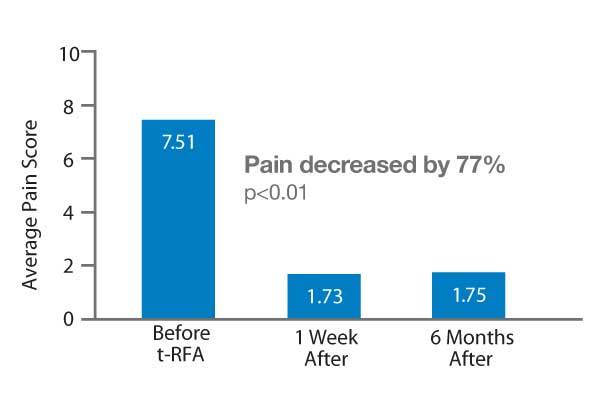
Clinical data show that rapid and lasting pain relief from metastatic spinal tumors is a reality for patients who receive t-RFA using the STAR System. This multicenter retrospective study included 128 treated lesions in 92 patients, many of whom had failed radiation therapy. Pain was decreased by 77% and 54% of the patients were able to reduce pain medication usage after t-RFA.
“If you look at a lot of the treatments that are used [to treat metastatic spine tumor pain], quality of life and life expectancy are not included. And they should be factored in. Daily we physicians see these patients suffering with pain that has dramatically affected the life they have left.”
Jack Jennings, Ph.D, M.D.*
Assistant Professor, Radiology
Washington University School of Medicine
Director of Musculoskeletal and Spine Interventions
Mallinckrodt Institute of Radiology
Targeted Radiofrequency Ablation(t-RFA) allows patients to concentrate on primary cancer treatments by quickly addressing painful metastatic lesions.
t-RFA is non-toxic, thereby allowing patients to continue their current systemic treatment uninterrupted.
The STAR™ System enables a single, minimally invasive procedure that can provide rapid, durable pain relief and localized tumor destruction.
The STAR™ System offers a treatment path for patients with limited options, including those with radio-resistant tumors or who have reached their radiation dose limits.
The SpineSTAR® Ablation Instrument is designed for use in the complex anatomy of the spine. This small steerable device is inserted into the vertebral body to deliver targeted RF energy to the tumor. RF energy heats and destroys metastatic tumor cells, while temperature sensors within the SpineSTAR® continuously monitors the ablation zone progress to minimize patient risk.
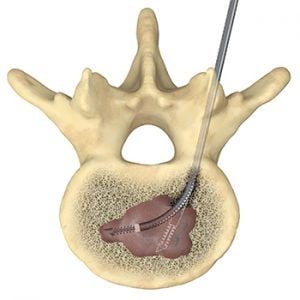
SpineSTAR® is placed in the tumor
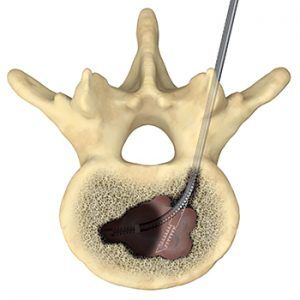
RF energy delivered
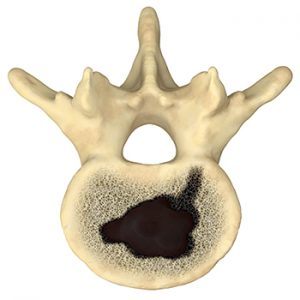
Tumor ablated
“There are a number of features of the [SpineSTAR®] device that I really like; the thermocouples on the tip allow me to actually measure [monitor real-time progress of] the ablation zone. The articulating tip allows and facilitates reaching to areas in the vertebrae that are not possible with a straight fixed device.”
Aaron Frodsham, M.D.
Interventional Radiology
University of Utah Huntsman Cancer Center
Salt Lake City, UT
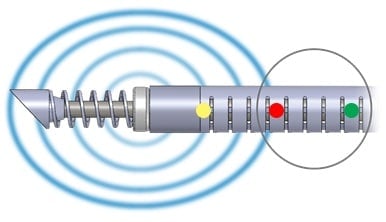
SpineSTAR® 5 | 10:
SpineSTAR® 10 | 15:
*Paid consultant of Merit Medical